I am looking for: Learn arrow down
I am looking for:
-
- Agriculture, Food & Natural Resources
- Architecture & Construction
- Arts, Audio/Video Technology & Communications
- Business Management & Administration
- Education & Training
- Finance
- Government & Public Administration
- Health Science
- Hospitality & Tourism
- Human Services
- Information Technology
- Law, Public Safety, Corrections & Security
- Manufacturing
- Marketing
- Science, Technology, Engineering & Mathematics
- Transportation, Distribution & Logistics
- View All
Categories
Categories
Search For
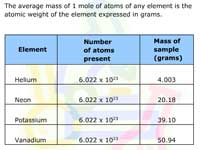
The Mole and Avogadro's Number
Learners examine how chemists use moles to "count" atoms by weight. Examples are given.
By Debbie McClinton Dr. Miriam Douglass Dr. Martin McClinton
Power Factor: A Mechanical Work Analogy
Learners read an analogy comparing mechanical work (in this case, sliding a weight) to that of electrical power. The relationship of work, apparent work, and power factor is developed.
By Roger Brown
Conversion Between Mass and Moles of an Element (Screencast)
Atomic weights are used to convert the mass of a sample into the number of moles of the element in the sample and vice versa. Four examples are provided for practice.
By Dr. Miriam Douglass Dr. Martin McClinton